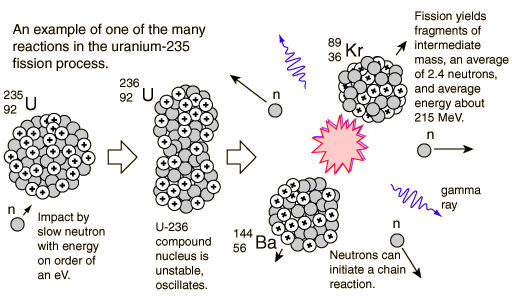 
While uranium-235 is the isotope that undergoes fission it is worth noting that uranium-238 atoms can absorb neutrons to become plutonium-239 which is another atom that can undergo fission. We will consider where the energy comes from in the next section. It is 50 million times more energy than burning the equivalent amount of carbon. The energy released per fission is relatively large.The neutrons may need to be slowed down and are then referred to as thermal neutrons. Both the number and speed of the neutrons is crucial within a working reactor. The Fission Process In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235. In a neutron flux, 235U, which is referred to as a fissile nuclide, absorbs neutrons and either fissions or forms 236U. For fission to occur the neutrons must be going at the right speed – too fast and they will bounce off rather than be absorbed.Once started, fission can become self-sustaining – this is called a chain reaction. The fact that neutrons are also produced means that these neutrons can go on to induce further fissions.Use data from the graph to show that the energy released as a result of. These by-products of nuclear power form the majority of the radioactive waste that we will consider next week. (a) A uranium-235, 235U, nucleus fissions into two approximately equally sized products. Fission products tend to be radioactive.In words this would be: ‘A uranium-235 atom absorbs a neutron to become uranium-236 which then undergoes fission to form the products xenon-140 and strontium-93 with three neutrons.’ An equation representing this particular fission would be: The fission products themselves can vary but examples would be xenon-140 and strontium-93. Neutrons are shown as using the same notation as for isotopes. The Centers for Disease Control and Prevention (CDC) protects people’s health and safety by preventing and controlling diseases and injuries enhances health decisions by providing credible information on critical health issues and promotes healthy living through strong partnerships with local, national, and international organizations.This image shoes the particles and types of radiation involved in fission Figure 14 The particles and types of radiation involved in fission įor more information on protecting yourself before or during a radiologic emergency, see CDC’s fact sheet titled “Frequently Asked Questions (FAQs) About a Radiation Emergency” at, and “Sheltering in Place During a Radiation Emergency,” at. Uranium is also a toxic chemical, meaning that ingestion of uranium can cause kidney damage from its chemical properties much sooner than its radioactive properties would cause cancers of the bone or liver.įor more information about U-235 and U-238, see the Public Health Statement by the Agency for Toxic Substances and Disease Registry at, or visit the Environmental Protection Agency at. Inhaling large concentrations of uranium can cause lung cancer from the exposure to alpha particles. Ingestion of high concentrations of uranium, however, can cause severe health effects, such as cancer of the bone or liver. 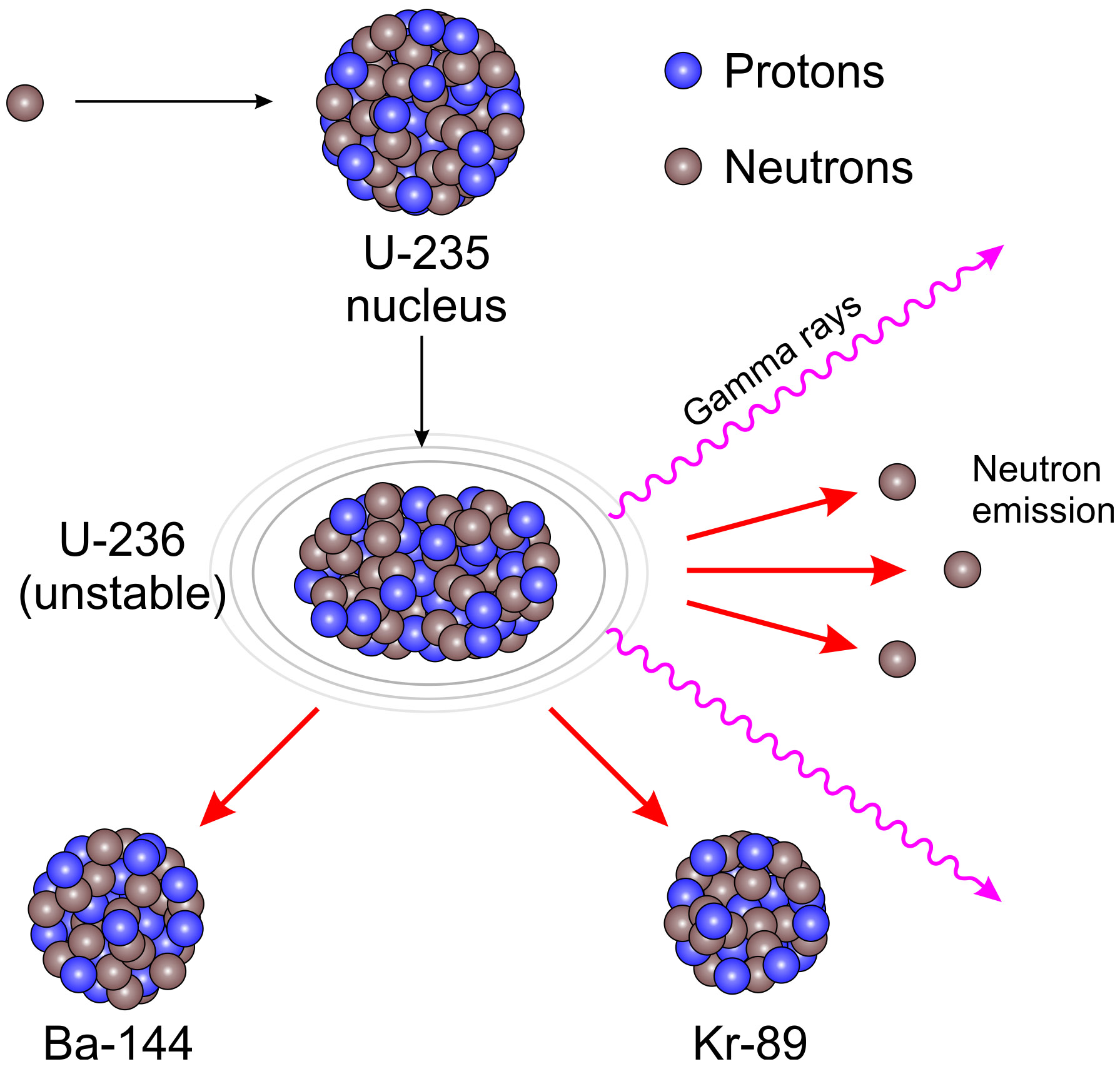
When it has been refined and enriched, uranium is a silvery-white metal.īecause uranium decays by alpha particles, external exposure to uranium is not as dangerous as exposure to other radioactive elements because the skin will block the alpha particles. Enriched uranium can be in the form of small pellets that are packaged in the long tubes used in nuclear reactors. U-235 can be concentrated in a process called “enrichment,” making it suitable for use in nuclear reactors or weapons. U-238 is the most abundant form in the environment. U-235 and U-238 occur naturally in nearly all rock, soil, and water. It also can be used in nuclear weapons.ĭepleted uranium (uranium containing mostly U-238) can be used for radiation shielding or as projectiles in armor-piercing weapons. Uranium “enriched” into U-235 concentrations can be used as fuel for nuclear power plants and the nuclear reactors that run naval ships and submarines. WILKINSON Nature 158, 163 ( 1946) Cite this article 2344 Accesses 36 Citations Metrics Abstract THE first quantitative measurements on the. During nuclear fission, a neutron collides with a uranium atom and splits it, releasing a large amount of energy in the form of heat and radiation. Chemical properties: Weakly radioactive, extremely dense metal (65% denser than lead)
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
